
400 MG Bevacizumab Injection
400 MG Bevacizumab Injection Specification
- Dosage Form
- Injection
- Salt Composition
- Bevacizumab 400 mg
- Indication
- Cancer (Colorectal, Lung, Glioblastoma, Renal Cell Carcinoma, Cervical Cancer, Ovarian Cancer)
- Feature
- Sterile and pyrogen-free, ready to use, single dose vial
- Ingredients
- Bevacizumab (concentrate for solution), excipients including trehalose, polysorbate 20, sodium phosphate, water for injection
- Application
- Intravenous infusion
- Ph Level
- 6.2
- Physical Color/Texture
- Clear to slightly opalescent, colorless to pale brown liquid
- Storage Instructions
- Dry place
- Shelf Life
- 24 Months
- Route of Administration
- Intravenous (IV) infusions only
- Prescription/Non Prescription
- Prescription
- Packaging Type
- Single-use vial
- Caution
- Administer under medical supervision only
- Strength
- 400 mg/16 mL (25 mg/mL)
400 MG Bevacizumab Injection Trade Information
- Minimum Order Quantity
- 1 Unit
- Payment Terms
- Cash Advance (CA)
- Supply Ability
- 5000 Units Per Month
- Delivery Time
- 5-7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About 400 MG Bevacizumab Injection
Expansive Therapeutic Area & Advanced Features
The 400 MG Bevacizumab Injection is primarily utilized for intravenous treatment of multiple malignant cancers, including colorectal, lung, glioblastoma, renal cell carcinoma, cervical, and ovarian cancer. As a ready-to-use single-dose vial, it prioritizes patient safety and convenience. Sterile, pyrogen-free, and featuring an optimized pH level of 6.2, it combines reliability and efficacy in every infused dose. Only prescribed and administered by professionals, it is tailored for use in advanced medical settings.
Sample Availability & Reliable Supply Protocols
We offer sample handover for quality assurance prior to bulk orders of the 400 MG Bevacizumab Injection. Secure payment terms are in place to ensure a smooth transaction. With robust supply ability, our goods are carefully dispatched and timely shipped, maintaining compliance with all pharmaceutical regulations. Every vial is managed with care from handover to delivery, solidifying our commitment to trusted, efficient pharmaceutical logistics.
FAQ's of 400 MG Bevacizumab Injection:
Q: How should the 400 MG Bevacizumab Injection be administered?
A: This injection must be given as an intravenous (IV) infusion under the direct supervision of a healthcare professional, strictly via prescription.Q: What types of cancer is this injection indicated for?
A: It is indicated for colorectal, lung, glioblastoma, renal cell carcinoma, cervical, and ovarian cancers, among others.Q: When should Bevacizumab Injection be stored and at what conditions?
A: Store the vial in a dry place under recommended storage conditions to maintain its efficacy throughout the 24-month shelf life.Q: Where is this product available for export or supply?
A: The 400 MG Bevacizumab Injection is available for export, supply, and trading, with operations based in India.Q: What are the key features of this injection formulation?
A: This product is a sterile, pyrogen-free concentrate that is ready-to-use, presented in a clear, colorless to pale brown, single-use vial.Q: What is the process of sample handover and shipping for bulk orders?
A: Samples can be provided before placing large orders. Shipped goods are dispatched promptly, ensuring compliance and efficient delivery.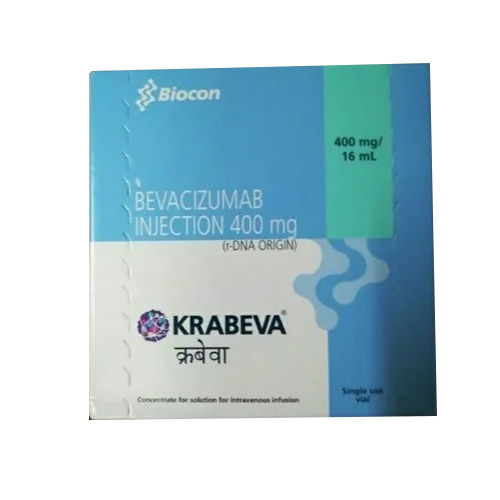

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
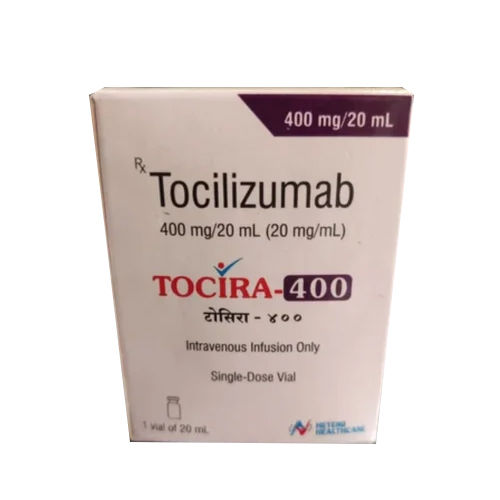
400 MG Tocilizumab Injection
Price 18000 INR / Box
Minimum Order Quantity : 10 Boxes
Dosage Form : Injection
Storage Instructions : Dry place
Salt Composition : Tocilizumab 400 mg
Indication : Rheumatoid Arthritis, Systemic Juvenile Idiopathic Arthritis, Cytokine Release Syndrome
80 MG Docetaxel Injection IP
Minimum Order Quantity : 10 Units
Dosage Form : Injection
Storage Instructions : Dry place
Salt Composition : Docetaxel 80 mg
Indication : Breast cancer, nonsmall cell lung cancer, prostate cancer, gastric cancer, head and neck cancer
Paclitaxel Injection IP
Minimum Order Quantity : 10 Units
Dosage Form : Injection
Storage Instructions : Dry place
Salt Composition : Paclitaxel
Indication : Used in the treatment of ovarian cancer, breast cancer, nonsmall cell lung cancer, and AIDSrelated Kaposis sarcoma.
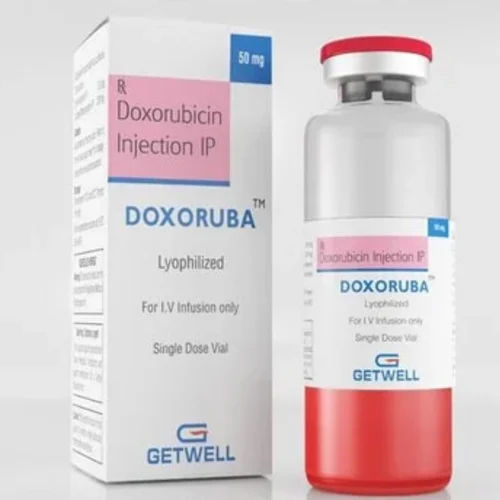
50 MG Doxorubicin Injection IP
Minimum Order Quantity : 10 Units
Dosage Form : Injection
Storage Instructions : Dry place
Salt Composition : Doxorubicin Hydrochloride 50 mg
Indication : Used for the treatment of various cancers including breast cancer, lymphomas, leukemias, and ovarian cancer.

 Send Inquiry
Send Inquiry




 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese