
Bortether 3.5 mg
Price 500 INR/ Vial
Bortether 3.5 mg Specification
- Origin of Medicine
- India
- Dosage Form
- Injection
- Life Span
- 24 months from manufacture date
- Salt Composition
- Bortezomib 3.5 mg
- Brand Name
- Bortether
- Indication
- Multiple Myeloma, Mantle Cell Lymphoma
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Packaging Type
- Glass vial with flip-off seal
- Drug Type
- Allopathic
- Ingredients
- Bortezomib
- Physical Form
- Lyophilized Powder for Injection
- Function
- Antineoplastic agent (proteasome inhibitor)
- Recommended For
- Adults with Multiple Myeloma or Mantle Cell Lymphoma
- Dosage
- As directed by physician; typically 1.3 mg/m2 body surface area
- Dosage Guidelines
- Use as prescribed by healthcare professional; intravenous or subcutaneous use only
- Suitable For
- Adults
- Quantity
- 1 Vial
- Storage Instructions
- Store below 25C, protect from light
- Marketed By
- Mylan
- Contraindications
- Hypersensitivity to bortezomib or boron
- Reconstitution Required
- Yes, with sterile normal saline solution
- Prescription Required
- Yes
- Schedule
- Schedule H (Prescription Drug)
- Route of Administration
- Intravenous or Subcutaneous
- Color
- White to off-white powder
- ATC Code
- L01XX32
- Side Effects
- Peripheral neuropathy, thrombocytopenia, fatigue, nausea
Bortether 3.5 mg Trade Information
- Minimum Order Quantity
- 10 Vials
- Payment Terms
- Cash Advance (CA)
- Supply Ability
- 5000 Vials Per Month
- Delivery Time
- 5-7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Bortether 3.5 mg
Special Features and Applications of Bortether 3.5 mg
Bortether 3.5 mg is specifically designed for adult patients battling Multiple Myeloma and Mantle Cell Lymphoma. Its use type is prescribed-only, ensuring safe administration under medical guidance. Apart from hematologic malignancies, its powerful proteasome inhibitor action extends to experimental treatments in oncology. Special features include a long shelf life of 24 months, easy reconstitution, and reliable efficacy, making it a sought-after choice among healthcare professionals and exporters in India.
Packaging, Sample Policy & Supply Details of Bortether 3.5 mg
Packing & Dispatch of Bortether 3.5 mg is carried out with utmost care: each glass vial is securely sealed and boxed for optimal safety. Transportation logistics are streamlined to ensure that stock is ready and supplied efficiently to buyers worldwide. Sample policies may be available upon request, and robust supply ability ensures consistent availability without interruptions, backed by top-tier warehousing and distribution channels.
FAQ's of Bortether 3.5 mg:
Q: How is Bortether 3.5 mg administered?
A: Bortether 3.5 mg is administered either intravenously or subcutaneously, and must be reconstituted with sterile normal saline solution before use. Administration should only be performed by a healthcare professional.Q: What conditions is Bortether 3.5 mg recommended for?
A: This medication is primarily recommended for adults diagnosed with Multiple Myeloma or Mantle Cell Lymphoma, as directed by a qualified physician.Q: When should I seek medical advice while using Bortether 3.5 mg?
A: If you experience unexpected side effects such as severe peripheral neuropathy, thrombocytopenia, or any signs of allergic reaction, contact your healthcare provider immediately for guidance.Q: Where should Bortether 3.5 mg be stored?
A: The vial should be stored below 25C, away from direct light. Always keep it in its original packaging until ready for use, following all storage recommendations provided by your pharmacist or healthcare provider.Q: What is the process for reconstituting Bortether 3.5 mg?
A: Bortether 3.5 mg must be reconstituted by a healthcare professional using sterile normal saline as per the product guidelines, ensuring the powder fully dissolves before administration.Q: How does Bortether 3.5 mg function in cancer treatment?
A: Bortether acts as a proteasome inhibitor, impeding protein degradation within cancer cells, thereby inhibiting tumor growth and enhancing the effectiveness of cancer therapies.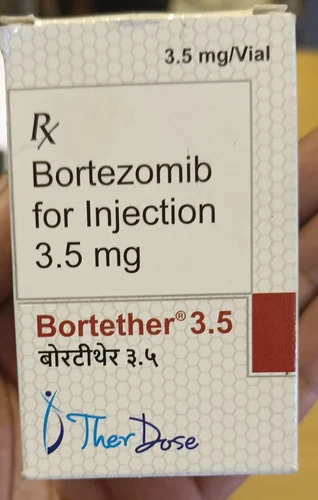

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Injection Category
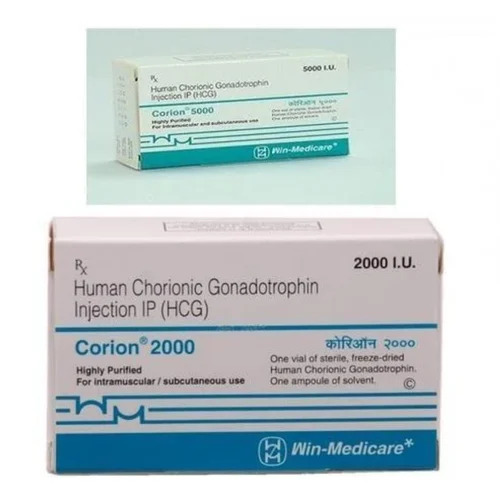
Chorionic Gonadotropin Injection 2000i.U.
Price 175 INR / Box
Minimum Order Quantity : 10 Boxes
Packaging Type : Glass Bottle
Dosage : As prescribed by physician
Dosage Guidelines : Injectable, follow physicians advice
Drug Type : Other, Allopathic
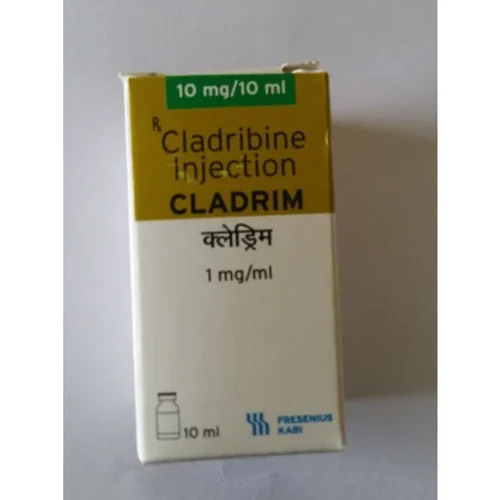
Cladrim Cladribine Injection
Price 9000 INR / Vial
Minimum Order Quantity : 10 Vials
Packaging Type : Vial
Dosage : As directed by physician
Dosage Guidelines : Follow medical advice
Drug Type : Other, Prescription
Erythropoietin Injacation 4000 Ml
Price 250 INR / Box
Minimum Order Quantity : 10 Boxes
Packaging Type : 1 x 1 ml, Injection
Dosage : As directed by the physician
Dosage Guidelines : Subcutaneous or intravenous use
Drug Type : Other, Biological
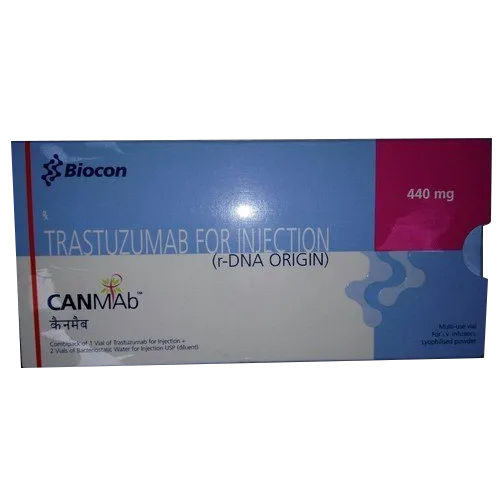
440 MG Trastuzumab For Injection
Price 20000 INR / Unit
Minimum Order Quantity : 10 Units
Dosage : As per instruction
Dosage Guidelines : As per required
Drug Type : Injection

 Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese