
Trastuzumab 440 Mg Injection
Price 15000 INR/ Box
Trastuzumab 440 Mg Injection Specification
- Packaging Type
- Vial
- Dosage Form
- Lyophilized powder for injection
- Origin of Medicine
- India
- Indication
- Metastatic breast, gastric cancer
- Life Span
- 24 months
- Brand Name
- CANMAB
- Salt Composition
- Trastuzumab
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Drug Type
- Biological
- Ingredients
- Trastuzumab
- Physical Form
- Injection
- Function
- Anticancer
- Recommended For
- HER2-positive breast cancer, gastric cancer
- Dosage
- As directed by the physician
- Dosage Guidelines
- Follow physicians instructions
- Suitable For
- Adults
- Quantity
- 1 Vial
- Storage Instructions
- Store at 2C-8C, do not freeze
- Prescription
- Prescription only
- Reconstitution Required
- Yes
- Route
- Intravenous
- Strength
- 440 mg
- HER2 Status
- Positive required
- Appearance
- White to off-white powder
- Diluent Included
- Yes
- Marketed By
- Biocon
- Dilution Required
- Yes
Trastuzumab 440 Mg Injection Trade Information
- Minimum Order Quantity
- 1 Box
- Supply Ability
- 1000 Boxes Per Month
- Delivery Time
- 7 Days
About Trastuzumab 440 Mg Injection
Outstanding Benefits & Advanced Features
CANMAB (Trastuzumab 440 Mg Injection) distinguishes itself with its rarefied composition, offering targeted action for HER2-positive cancers. Ready-to-reconstitute powder, supplied with diluent, ensures precise dosing. Backed by Biocon's champion manufacturing, each vial promises a 24-month shelf life with optimal storage at 2-8C. Tamper-proof packaging and stringent quality assurance make CANMAB a true first-class option for healthcare institutions and professionals focusing on advanced cancer therapies.
Domestic & Export Market Coverage
Trastuzumab 440 Mg Injection enjoys strong rates across India's main domestic markets and is frequently supplied from major FOB ports. Each single vial per box is delivered with meticulous packaging to ensure product integrity throughout transit. As a leading exporter, supplier, and trader from India, we keep expenditure competitive for both local institutions and the export market, making CANMAB an attractive therapeutic investment worldwide.
FAQ's of Trastuzumab 440 Mg Injection:
Q: How should Trastuzumab 440 Mg Injection be stored?
A: Trastuzumab 440 Mg Injection must be stored between 2C and 8C. Ensure that it is not frozen and keep it in its original packaging until use to maintain its potency and stability.Q: What is the process for preparing this injection?
A: The injection is supplied as a lyophilized powder and requires both reconstitution and dilution. Carefully follow the provided instructions using the included diluent, and administer intravenously as directed by a healthcare professional.Q: When is Trastuzumab 440 Mg Injection recommended?
A: This injection is indicated for adults with HER2-positive metastatic breast or gastric cancer. The dosage and treatment schedule are determined by the prescribing physician based on patient needs and cancer progression.Q: Where is CANMAB (Trastuzumab) primarily marketed and exported from?
A: CANMAB is mainly marketed across India and exported globally through major Indian ports. The product consistently meets high demand among hospitals, clinics, and cancer treatment centers in both domestic and international markets.Q: What are the primary benefits of choosing CANMAB over alternatives?
A: CANMAB offers a champion combination of targeted efficacy, first-class manufacturing quality, long shelf life, and thoughtfully designed packaging. Backed by Biocon's reputation, it provides reliable results for HER2-positive cancer therapy.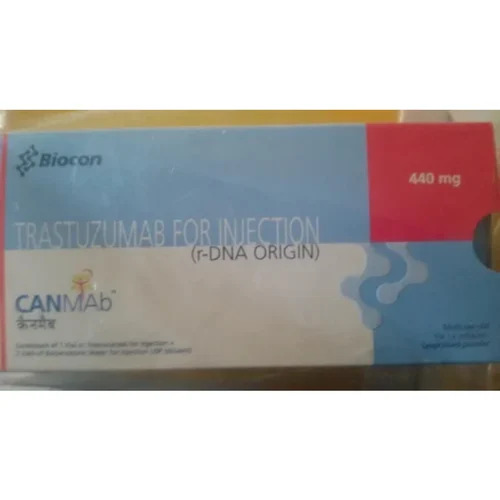

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Injections Category
CS - II Forte 1.5 gm (Cefoperazone , Sa)
Price 324 INR / Vial
Minimum Order Quantity : 10 Vials
Dosage : As directed by physician
Physical Form : Other, Powder for Injection
Drug Type : Other, Antibiotic
Indication : Serious bacterial infections
Alprostadil Injection Prostaver 500 mcg
Price 5650 INR / Piece
Minimum Order Quantity : 1 Piece
Dosage : As directed by physician
Physical Form : Liquid
Drug Type : Other, Allopathic
Indication : Erectile dysfunction
Taxonab (Paclitaxel)
Price 900 INR / Vial
Minimum Order Quantity : 1 Vial
Dosage : As directed by physician
Physical Form : Other, Injection
Drug Type : Other, Allopathic
Indication : Breast, ovarian, lung cancer
Zeroklot 40 Mg0.4 Ml Enoxaparin Injection
Price 363 INR / Box
Minimum Order Quantity : 10 Boxes
Dosage : 40 mg/0.4 ml
Physical Form : Other, Injection
Drug Type : Other, Anticoagulant Drug
Indication : Prevention and treatment of blood clots

 Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese